Author: Noah Noonan, BS
Clinical Perspective: John D. Powderly MD, CPI
Advancements During a Pandemic
The COVID-19 pandemic has radically interrupted the American healthcare landscape in a manner unseen since the 2009 H1N1 Influenza Pandemic (CDC). Now more than ever, healthcare, pharmaceuticals, and biotechnology are in the public spotlight as the world tenuously awaits a vaccine. This is an opportune moment to reflect on how these fields have collectively advanced the treatment of cancer, prolonged the life of cancer patients, and improved the quality of life for those afflicted. Cancer patients must now confront a new challenge: fighting their disease during a public health crisis. Fortunately, groundbreaking cancer research has continued even in the face of global supply chain breakdowns and disruptions to the workforce, and the field of Immuno-Oncology continues to advance.
Cancer Immunotherapy, or Immuno-oncology, involves the use of checkpoint inhibitors, cellular therapy, cancer vaccines, and other modalities to treat cancer. Immuno-oncology is now an established and effective treatment option for both solid and hematological malignancies. The field can be traced back over 100 years ago to anecdotal reports of systemic anti-tumor response after irradiation of only a single tumor lesion or administration of Coley Toxins – intentional injections of infectious bacteria designed to stimulate the immune system. Additionally, many drugs developed through the late 20th century are immunotherapeutics, even if the mechanisms of action were unknown at the time. While Immuno-oncology encompasses many drug classes, the main drivers for this field have been checkpoint inhibitors and cellular therapies [1].
 The first wave of immuno-oncology trial approvals arrived in the early 2010’s and proceeded to undergo massive growth. From 2014 to 2017, new trials in immuno-oncology soared from 250 to almost 600, reaching double digit growth rates each year as drug classes diversified. This rapid growth was mostly driven by biomarker and checkpoint inhibitor studies. Biomarker studies seek to correlate measurements of substances in the body to the occurrence and outcome of a disease or health event. Checkpoint inhibitor studies are designed to target regulating mechanisms within the immune system that can either increase or decrease the bodies response to infection, insult, and injury. In 2011, checkpoint inhibitor ipilimumab received a seminal U.S. Food and Drug Administration (FDA) approval for the treatment of melanoma. Ipilimumab targets cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), a protein that down regulates the immune response, and became the first checkpoint inhibitor to be approved by the FDA. Dr. James P. Allison shared the Nobel Prize in Physiology or Medicine in 2018 for his discovery of the CLTA-4 pathway and its involvement in cancer immunotherapy. In 2014 the first programmed cell death protein 1 (PD-1) inhibitor, nivolumab, was approved by the FDA. The PD-1/PD-L1 pathway has since become a staple in field of Immuno-oncology due to its strong modulation of the T cell response. John D. Powderly MD, CPI, founder and president of Carolina BioOncology Institute, was a Principal Investigator in the early phase 1 trials for CTLA-4, PD1, and PDL1 mAbs.
The first wave of immuno-oncology trial approvals arrived in the early 2010’s and proceeded to undergo massive growth. From 2014 to 2017, new trials in immuno-oncology soared from 250 to almost 600, reaching double digit growth rates each year as drug classes diversified. This rapid growth was mostly driven by biomarker and checkpoint inhibitor studies. Biomarker studies seek to correlate measurements of substances in the body to the occurrence and outcome of a disease or health event. Checkpoint inhibitor studies are designed to target regulating mechanisms within the immune system that can either increase or decrease the bodies response to infection, insult, and injury. In 2011, checkpoint inhibitor ipilimumab received a seminal U.S. Food and Drug Administration (FDA) approval for the treatment of melanoma. Ipilimumab targets cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), a protein that down regulates the immune response, and became the first checkpoint inhibitor to be approved by the FDA. Dr. James P. Allison shared the Nobel Prize in Physiology or Medicine in 2018 for his discovery of the CLTA-4 pathway and its involvement in cancer immunotherapy. In 2014 the first programmed cell death protein 1 (PD-1) inhibitor, nivolumab, was approved by the FDA. The PD-1/PD-L1 pathway has since become a staple in field of Immuno-oncology due to its strong modulation of the T cell response. John D. Powderly MD, CPI, founder and president of Carolina BioOncology Institute, was a Principal Investigator in the early phase 1 trials for CTLA-4, PD1, and PDL1 mAbs.
Contrary to the previous decade of growth, there has been a notable plateau in the number of Immuno-Oncology approvals into 2019 [2]. This indicates both a maturation of the market and the patient population which these therapeutics target. The evidence for a maturing market is corroborated by a statistical analysis performed with a literature mining approach. This literature mining analysis was generated from a pool of over 93,000 publications in the field of Immuno-Oncology and showed a clear transition from early phase clinical trials to randomized control trials in 2019-2020. The analysis also noted that the most prevalent cancer types included in these studies were breast, lung, and colorectal and observed a shift in focus from hematological to solid tumors [5].
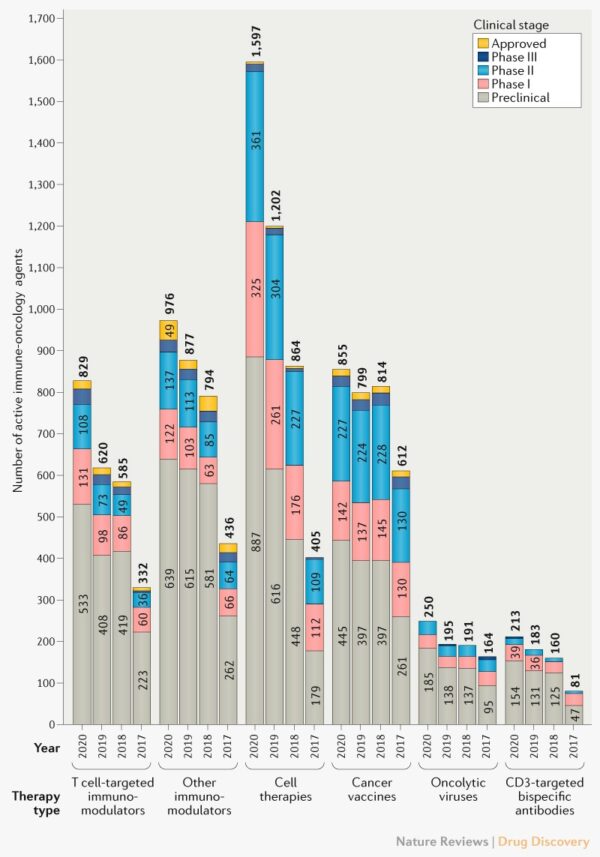
While the Immuno-Oncology market may be maturing in certain drug classes, cell therapies are growing rapidly and outpacing all other immunotherapeutics. Globally, the cancer cell therapy pipeline included 1,483 active agents in March 2020 — an increase of 472 agents from March 2019. Chimeric antigen receptor T (CAR-T) cell products saw the greatest growth during this time (77% increase from 2019 to 2020) [9]. A recent article published by Upadhaya et al. in Nature Reviews Drug Discovery (202) illustrates how in just five months, as of August 14, 2020, the number of cancer cell therapy agents has increased by 7.7% to 1,597 (figure above left) [8]. This increase in cell therapy agents under study is also shaping the physical Immuno-Oncology landscape in regional distribution of development. The initial wave of checkpoint inhibitor studies came mostly from the US and into Europe. Now, alongside the United States and Europe, China is also a large contributor to cell therapy investigational products [1].
A Maturing Market with Room to Grow
Although there are indicators of the checkpoint inhibitor market reaching maturity, the prospect of future research in Immuno-Oncology is promising. Expanding the patient populations for clinical trials, exploring combination therapies, and deciphering greater knowledge of biomarkers all have the potential to open more doors for drug discovery.
Patient recruitment and the intricacies of running clinical trials could be limiting to the number of new trials being initiated. A review of 13 studies published in the Journal of the National Cancer Institute (JNCI) showed that around 8% of cancer patients participate in clinical trials while 75% of patients wishing to participate are not able. The publication argues that the reason for low participation in clinical trials is not only barriers to patient decisions, but also structural barriers in their execution[7]. Overly strict eligibility criteria, such as requiring patients to have failed standard chemo, and unmet needs of certain tumor types, prevent many patients from enrolling in trials. The availability of trial sites for patients is also decreased by administrative burdens and higher potential revenue for a buy-and-bill standard of care model as opposed to clinical trials. Addressing these factors could make trials more accessible to the public and open more opportunities for the checkpoint inhibitor market.
There is further opportunity for expansion in checkpoint inhibitor research through combinational approaches. Currently, there are 170 studies being conducted on the efficacy of chemotherapy in combination with PD-1 and PD-L1 blockades [6]. Other studies are investigating the effect of radiotherapy or surgery in combination with checkpoint inhibitors. One randomized phase I study investigated the effects of nivolumab and ipilimumab as neoadjuvant therapy for resectable melanoma and found increases in tumor specific T-cell clones when compared to adjuvant treatment — evidence for immune response priming prior to surgery [3]. A separate study on neoadjuvant immunotherapy treatment in microsatellite instable colorectal cancer found a response rate in all 7 patients, and a complete response in 4 out of 7 patients [4]. In addition to combination with other standards of care, there are countless possibilities for the combination of multiple immunotherapy agents.
The National Cancer Institute (NCI) defines a biomarker as a biological molecule found in blood, other body fluids, or tissues that is a sign of a normal or abnormal process, or of a condition or disease Biomarker profiling can help oncologists make more informed decisions when considering a patients’ best course of treatment. Monitoring biomarkers during cancer patients’ treatment can empower physicians to predict response more accurately. Research is currently being conducted on the feasibility and accuracy of gathering biomarker information from patient peripheral blood as opposed to tumor tissue. The advantages of using peripheral blood draws as a biomarker source include increased patient safety, decreased invasiveness, and an ability to repeat biomarker analysis at a higher frequency.
Two biomarkers that have gained significant interest are circulating tumor DNA (ctDNA) and Circulating Tumor Cells (CTC). Both biomarkers can offer value to patients by providing patient-specific information to physicians. However, while ctDNA can help reveal which mutations define the tumor mutanome, it does not provide information about the proteins, or expression of druggable targets for a tumor. Conversely, CTCs are the biomarker of the future because they can help investigators identify druggable targets such as tumor checkpoint expression and/or immune escape mechanisms which are not clearly identifiable in ctDNA. Additionally, CTCs can provide prognostic information when enumerated in whole peripheral blood of cancer patients. Furthermore, viable CTCs can potentially be isolated and cultured, thus become a source of tumor-associated antigens (TAA) to produce personalized immunotherapies. Future personalized medicine may depend upon ctDNA in conjunction with viable ex-vivo cultured CTCs. CBOI & BioCytics Human Applications Lab are one the few research groups in the United States focused on enumerating, profiling, and culturing CTCs from leukapheresis towards personalized immunotherapies.
From 2019 to 2020, a concerted effort investigated the predictability of tumor biomarkers like tumor microsatellite instability (MSI), tumor mutational burden (TMB), and blood-soluble tumor biomarkers, including ctDNA. Further research, potentially from retrospective data review, may help predict immunotherapy response in conjunction with associated tumor biomarkers. Preclinical research also has the potential to discover new biomarkers that may yield new druggable targets and pathways for therapy.
The Up and Coming
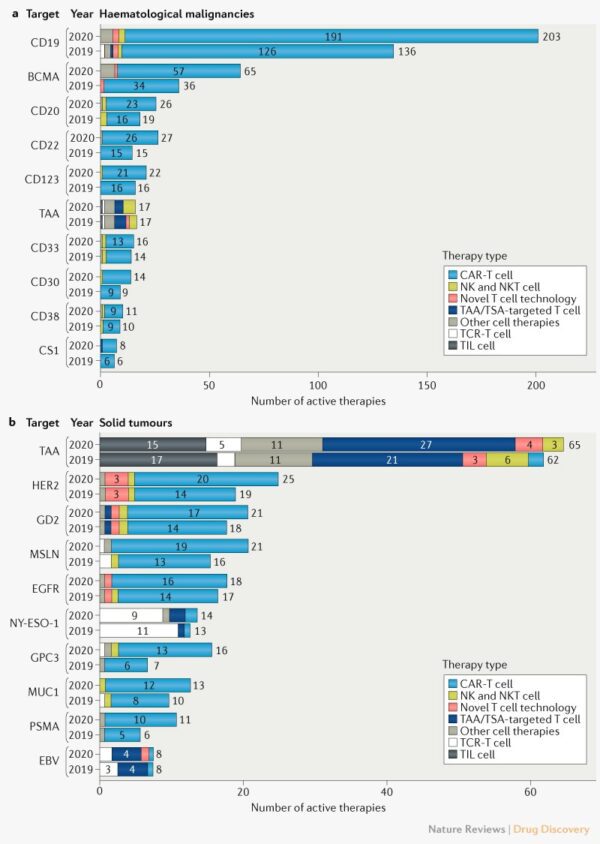 Cell therapy — transplantation of human cells to replace or repair damaged tissue and/or cells — is currently in the initial stages of discovery that checkpoint inhibitors were in 2014. As of August 2020, there were 1,597 cell therapy clinical trials alone [8]. The greatest increase in studies during this year came from preclinical and phase I studies of allogeneic therapies, those that come from a matched or unrelated donor, which require significant time and effort before adoption as viable treatment options. Interestingly, allogeneic T cell therapies have demonstrated greater efficacy in hematological cancers than solid tumors [9]. The B cell surface antigen CD19 remains the most targeted tumor antigen for hematological cancers. As shown in the figure on the right from Yu et al. published in Nature Reviews Drug Discovery (2020), most cell therapies in development for solid tumors target tumor-associated antigens such as HER2, GD2, mesothelin (MSLN), EGFR, NY-ESO-1, MUC1, and other undisclosed TAAs [9-10].
Cell therapy — transplantation of human cells to replace or repair damaged tissue and/or cells — is currently in the initial stages of discovery that checkpoint inhibitors were in 2014. As of August 2020, there were 1,597 cell therapy clinical trials alone [8]. The greatest increase in studies during this year came from preclinical and phase I studies of allogeneic therapies, those that come from a matched or unrelated donor, which require significant time and effort before adoption as viable treatment options. Interestingly, allogeneic T cell therapies have demonstrated greater efficacy in hematological cancers than solid tumors [9]. The B cell surface antigen CD19 remains the most targeted tumor antigen for hematological cancers. As shown in the figure on the right from Yu et al. published in Nature Reviews Drug Discovery (2020), most cell therapies in development for solid tumors target tumor-associated antigens such as HER2, GD2, mesothelin (MSLN), EGFR, NY-ESO-1, MUC1, and other undisclosed TAAs [9-10].
Historically, CAR-T studies have focused on hematological cancers because they have a well-defined public antigen, such as CD19. Unfortunately, investigators have found several challenges in translating the efficacy of CAR-T therapy from hematological cancers to solid tumors. This is due to many factors, but chief among these is vast differences in the tumor microenvironments (TME) of hematological cancers and solid tumors, a high number of potentially mutated tumor specific antigens in solid tumors, and difficulties in trafficking to and penetrating solid tumors. [1]. Potential solutions to create a more diverse repertoire of anti-tumor T-Cells include engineering an ex-vivo lymph node. Utilizing the endogenous tumor-reactive T cells circumvents the need for costly CAR-T or TCR engineering and may reduce the manufacturing process, chain of custody, and cost to the patient.
A Pathway Forward
Immuno-oncology is a field that has expanded rapidly in the last decade. In the last year, we have seen a maturation in the market of checkpoint inhibitors, new knowledge gained from biomarker statuses, and exponential growth in the field of cell therapy. With a maturing checkpoint inhibitor market, it is important to see an expansion of targeted patient populations, and to address other barriers to entry such as eligibility criteria and access to clinical trials. This market will continue to expand and find new forms of treatment through combinations with radiotherapy, chemotherapy, and surgery. As more retrospective reviews of biomarkers come from ongoing clinical trials, correlations can be made to response to various lines of therapy. As preclinical research finds new pathways and biomarkers, we will continue to see more novel drugs developed and brought to market. Finally, with the flourishing new market of cell therapy, challenges will be met to treat solid malignancies through genetically modified or endogenous tumor-reactive T-cells expanded ex-vivo. With the field of immunotherapy now established as pillar of treatment, cancer patients can find new hope in treatment options as the field continues to evolve.
- Altmann, D., TF. Gajewski, H., J. Gong, A., J. Tang, A., L. Apetoh, F., WM. Liu, D., . . . N. Zacharakis, H. (2019, June 19). Advances in cancer immunotherapy 2019 – latest trends. Retrieved August 28, 2020, from https://jeccr.biomedcentral.com/articles/10.1186/s13046-019-1266-0
- A 2020 Look at 3 Trends in Immuno-Oncology Biomarkers. (2020, March 09). Retrieved August 28, 2020, from https://www.decibio.com/2020/03/02/a-2020-look-at-3-trends-in-immuno-oncology-biomarkers/
- Blank CU, Rozeman EA, Fanchi LF, Sikorska K, van de Wiel B, Kvistborg P, et al. Neoadjuvant versus adjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma. Nat Med. 2018.
- Chalabi M, Fanchi LF, Van den Berg JG, Beets GL, Lopez-Yurda M, Aalbers AG, et al. LBA37_PRNeoadjuvant ipilimumab plus nivolumab in early stage colon cancer. Ann Oncol. 2018;29(suppl_8):mdy424.047-mdy424.047.
- Pouliliou, S., Nikolaidis, C. & Drosatos, G. Current trends in cancer immunotherapy: a literature-mining analysis. Cancer Immunol Immunother (2020). https://doi.org/10.1007/s00262-020-02630-8
- Tang J, Shalabi A, Hubbard-Lucey VM. Comprehensive analysis of the clinical immuno-oncology landscape. Ann Oncol. 2018;29(1):84–91.
- Unger JM, Vaidya R, Hershman DL, Minasian LM, Fleury ME. (2019, March) Systematic Review and Meta-Analysis of the Magnitude of Structural, Clinical, and Physician and Patient Barriers to Cancer Clinical Trial Participation, JNCI: Journal of the National Cancer Institute, Volume 111, Issue 3, Pages 245-255, https://doi.org/10.1093/jnci/djy221
- Upadhaya, S., Hubbard-Lucey, V. M., & Yu, J. X. (2020). Immuno-oncology drug development forges on despite COVID-19. Nature Reviews Drug Discovery. doi:10.1038/d41573-020-00166-1
- Yu, J. X., Upadhaya, S., Tatake, R., Barkalow, F., & Hubbard-Lucey, V. M. (2020). Cancer cell therapies: The clinical trial landscape. Nature Reviews Drug Discovery, 19(9), 583-584. doi:10.1038/d41573-020-00099-9
- Wagner, J., Wickman, E., Derenzo, C., & Gottschalk, S. (2020). CAR T Cell Therapy for Solid Tumors: Bright Future or Dark Reality? Molecular Therapy, 28(11), 2320-2339. doi:10.1016/j.ymthe.2020.09.015

