
To complement our Clinic’s on site Clinical Lab and Pharmacy, HAL research facilities are fully equipped with a diverse array of instruments for cell enrichment, analysis, expansion & culture, and sample preservation. HAL staff will work with each sponsor to ensure your protocol, assays, and preclinical product are developed on the platform of your choosing. All devices are user-configurable and our staff scientists, cell processing specialists, and research team will develop a custom plan for your research needs.

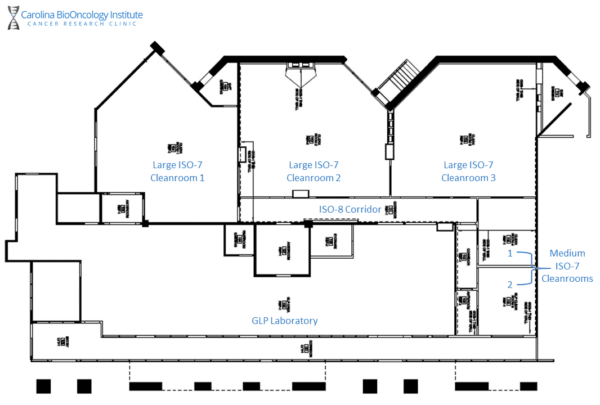
Our Cleanroom Facility
Our cleanroom facility is designed to provide flexible space for the development and manufacturing of your preclinical cell therapy products. It is comprised of three large ISO-7 cleanrooms and two medium ISO-7 cleanrooms. These units are built around a central ISO-8 hallway connecting the spaces via a dedicated anteroom and gowning area. The medium cleanrooms are also capable of both positive and negative pressure as dictated by process and sponsor needs.
The cleanroom facility is designed with architectural features and finishes in accordance with US FDA guidance and regulations as well as international requirements for controlled cleanroom manufacturing environments and aseptic processing:
- Floors and other exposed surfaces are designed to withstand the necessary cleaning and sanitization that ensure product purity and patient safety
- HEPA filter and air-handling units are configured to deliver particulate-free air; the air quality meets Class 10,000 (ISO class 7) standards
- Segregated HVAC equipment are built from best of breed equipment
- Critical parameters (temperature, pressure, and humidity) are continually monitored
- Environmental monitoring is performed via equipment also used by both the NCI and NIH
- All manufacturing equipment and utilities are validated in accordance with bio-pharmaceutical standards (IQ/OQ/PQ)
- Standard operating procedures assure that all systems are properly maintained and functional
- Access to the facility is keycard controlled
In addition to the cleanrooms, the facilities contain separate USP797/800 pharmacies, a dedicated shipping and receiving area and a large, contiguous GLP lab adjacent to the cleanrooms.





